  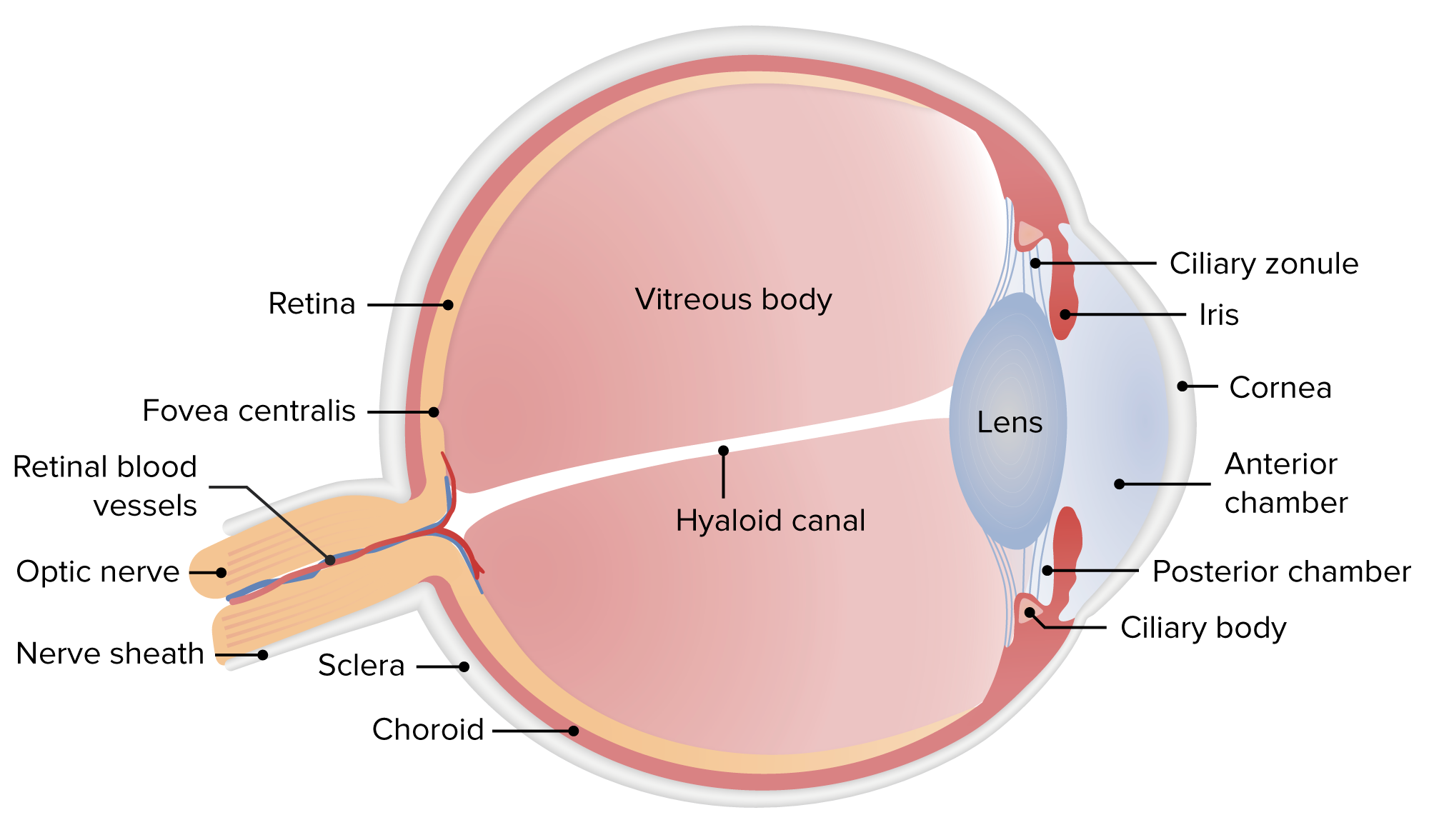
2017), which is related to its high expression in skin. MFSD12 was shown to be involved in pigmentation ( Crawford et al. Taken together, these results suggest that lysosome-localized MFSD12 may affect GSL metabolism. Consistent with the previous report ( Fukuda 1991), the C-terminus of LAMP1 faces the cytoplasm and its N-terminus faces the lysosome lumen ( Fig. As a positive control, we checked the orientation of LAMP1 by the same assay. S1I, both the FLAG and HA signals were detected in unpermeabilized and permeabilized lysosomes, indicating that both the N-terminus and C-terminus of MFSD12 are exposed to the cytoplasmic side. Lysosomes were purified from HEK293T cells with FLAG-MFSD12-HA expression, and the fluorescence signals in unpermeabilized and Triton X-100-permeabilized lysosomes were detected by flow cytometry. To investigate the transmembrane orientation of MFSD12, we made an MFSD12 construct with a FLAG-tag at its N-terminus and an HA-tag at its C-terminus. Similar to the cellular localization, MFSD12 was localized at the lysosomal membrane in the intestine ( Fig. To further confirm the localization of MFSD12 in tissues, we generated MFSD12-GFP knock-in mice ( Fig. S1F), which is consistent with the previous report ( Crawford et al. Immunostaining results showed that MFSD12 was co-localized with the lysosomal integral membrane protein LAMP1 ( Fig. S1D and S1E), indicating that MFSD12 may have an important function in these tissues. MFSD12 was highly expressed in the intestine and skin ( Fig. To explore the possible roles of MFSD12 in GSL metabolism, we tested the expression of MFSD12 in different adult mouse tissues by quantitative PCR (qPCR) and immunoblot ( Fig. These results suggest that MFSD12 may modulate GSL metabolism via regulation of GLB. 2020), increased GLB1-Luc activity almost 3-fold ( Fig. Surprisingly, knockdown of MFSD12, a gene involved in pigmentation and cysteine transport ( Crawford et al. We performed this screen twice with very good reproducibility (Spearman R = 0.98) and identified 12 genes that increased GLB1c-Luc activity by more than 2-fold ( Fig. The cells were transfected with a siRNA library targeting 82 human lysosomal transporter-encoding genes and a control vector, RSV-Luc, to monitor transfection efficiency and cell viability ( Fig. GLB1-Luc consists of a luciferase reporter gene linked to the promoter of the β-galactosidase gene GLB1. We established stable expression of the reporter GLB1-Luc in the human intestinal epithelial cell line HIEC-6. To identify which lysosomal transporters are involved in GSL metabolism, we tested the effect of 82 lysosomal membrane-localized transporters on expression of β-galactosidase (GLB1), an enzyme in GSL catabolism ( Hannun and Obeid 2018 Breiden and Sandhoff 2019) ( Fig. Although many genes are known to play critical roles in lysosomal homeostasis, it is still unclear whether they affect GSL metabolism. TFEB, a downstream target of mTOR and one of microphthalmia family of transcription factors, shuttles between the nucleus and cytoplasm to control the expression of genes required for lysosome biogenesis and autophagy ( Settembre et al. mTOR, a nutrient and energy sensor, regulates lysosomal function via transcription factor EB (TFEB)-mediated gene expression ( Settembre et al. The internal environment of the lysosome is controlled by lysosomal membrane proteins, such as vacuolar-type H +-ATPase (V-ATPase), which actively pump hydrogen ions into the lysosome to maintain the highly acidic environment within the lysosomal lumen for normal catabolism ( Mindell 2012). The lysosome is a central organelle for catabolic function in energy metabolism, nutrient sensing, and nutrient recycling in response to nutritional stress, and homeostatic mechanisms are critical for maintaining the catabolic function of lysosomes ( Mizushima and Komatsu 2011 Settembre et al. 
However, the non-lumenal lysosomal membrane proteins involved in GSL metabolism remain to be identified. However, in some LSDs, the inherited mutations affect other proteins, such as those residing in the lysosomal membrane these mutations are associated with defects in trafficking and fusion in the endocytic system ( Platt et al. The primary LSDs occur through the well-known mechanism of inherited defects in genes encoding the catabolic enzymes (or their related cofactors) which reside in the lumen of lysosomes ( Hannun and Obeid 2018 Breiden and Sandhoff 2019). Impairments of the catabolic pathway cause the onset of lysosomal storage diseases (LSDs), which are characterized by the aberrant accumulation of GSLs ( Hannun and Obeid 2018 Breiden and Sandhoff 2019). Glycosphingolipid (GSL) catabolism is strictly regulated in a sequential manner by lysosomal hydrolases with the help of lipid-binding proteins. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
